
Recently, a collaborative study led by Professor Dengfeng Cheng (Shanghai Clinical Research and Trial Center & the School of Biomedical Engineering, ShanghaiTech University) and Professor Zonghua Luo was published in Acta Pharmaceutica Sinica B (IF 14.6), a leading journal in pharmaceutical sciences. Their article, entitled “Preclinical evaluation of a novel tetrazine probe within a pretargeted delivery system for theranostics in HER2-positive tumor-bearing mice models”, presents a bioorthogonal click chemistry-based pretargeted radionuclide delivery system for HER2-positive tumors. This work provides a new strategy for high-contrast molecular imaging and safe, effective therapy of HER2-positive solid tumors.

HER2 is an important molecular target in a variety of solid tumors. Although HER2-targeted nuclear theranostics has advanced considerably in recent years, conventional approaches based on direct radiolabeling of antibodies still face several challenges, including prolonged blood circulation, slow clearance from non-target tissues, and relatively high radiation exposure to normal organs. These limitations have hindered the broader translational application of such strategies. To address these issues, the research team developed an inverse electron-demand Diels-Alder (IEDDA)-mediated pretargeting strategy (Figure 1). Unlike conventional direct-labeling approaches, this strategy first delivers a modified antibody to the tumor site. After sufficient accumulation in the tumor and gradual clearance from non-target tissues, a radiolabeled tetrazine-based small-molecule probe is administered. The probe then rapidly reacts with the prelocalized antibody through bioorthogonal click chemistry, enabling precise deposition of radioactive signals within the tumor. Compared with directly radiolabeled antibodies, this sequential strategy has the potential to reduce unnecessary radiation exposure to normal tissues while improving tumor-to-background contrast and therapeutic safety.
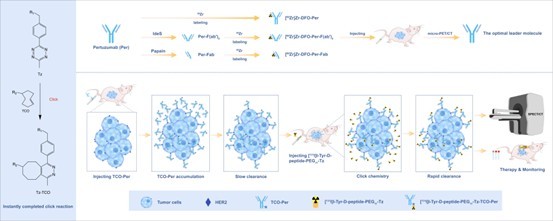
Figure 1. Schematic illustration of the HER2-targeted pretargeted theranostic system based on the tetrazine probe.
A key innovation of this study is the design and development of the novel tetrazine probe [131I]I-Tyr-D-peptide-PEG11-Tz. As the core effector molecule in the pretargeted system, the tetrazine group plays a decisive role in click reaction efficiency, in vivo stability, and overall delivery performance. To optimize these properties, the team systematically refined the probe with regard to reactivity, biodistribution, and in vivo applicability, ultimately constructing a new Tz probe with favorable characteristics for pretargeted theranostics. Structurally, the precursor of the probe consists of three major components (Figure 2): a tetrazine moiety for rapid ligation with the TCO-modified antibody, a PEG11 linker and D-peptide sequence to improve hydrophilicity and in vivo behavior, and a tyrosine residue for radioiodination. This integrated molecular design supports efficient click reactivity while maintaining radiolabeling feasibility, in vivo stability, and favorable pharmacokinetic properties, thereby providing an important foundation for the practical implementation of the pretargeting platform.
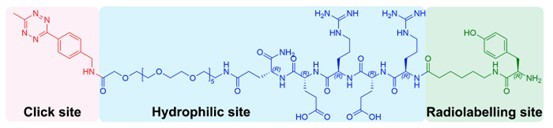
Figure 2. Design of the precursor of the novel tetrazine probe Tyr-D-peptide-PEG11-Tz.
The research team further performed lead antibody screening and optimization of pretargeting conditions, and subsequently evaluated the system in HER2-positive tumor-bearing models (Figure 3). The results showed that the novel Tz probe enabled effective tumor-targeted delivery within the pretargeted system and demonstrated rapid background clearance, high-contrast imaging performance, and promising therapeutic potential. These findings highlight the advantages of pretargeting strategies in HER2-targeted nuclear theranostics. More importantly, the study indicates that optimization of the tetrazine probe is not merely a technical refinement, but a critical determinant of the overall performance of the theranostic platform. Continued development of such probes may further improve the delivery efficiency and biosafety of radiopharmaceuticals in vivo, offering new tools and new directions for precision nuclear medicine in HER2-positive tumors and potentially in a wider range of solid malignancies.
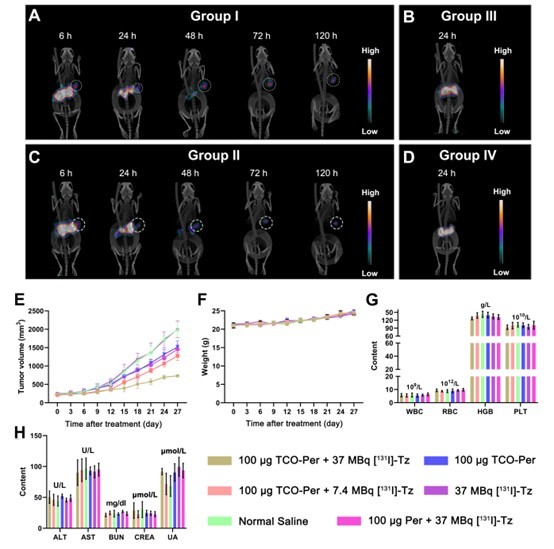
Figure 3. Imaging and therapeutic performance of the novel tetrazine probe in HER2-positive tumor-bearing models.
Overall, this study establishes and validates a novel HER2-targeted pretargeted theranostic platform with promising potential for both imaging and therapy. The work provides new experimental evidence supporting the application of bioorthogonal click chemistry in nuclear medicine and lays a foundation for the further optimization and future clinical translation of pretargeted theranostic strategies.
Professor Dengfeng Cheng from the Department of Nuclear Medicine, Shanghai Clinical Research and Trial Center, Dr. Dai Shi from the Department of Nuclear Medicine, Zhongshan Hospital, Fudan University, and Professor Zonghua Luo from the School of Biomedical Engineering, ShanghaiTech University are the co-corresponding authors of the study. Pengcheng Ma, a PhD candidate at Zhongshan Hospital, Fudan University, Associate Chief Physician Hui Tan from the Department of Nuclear Medicine, Zhongshan Hospital, Fudan University, and Associate Researcher Qingyu Lin from the Department of Nuclear Medicine, Shanghai Clinical Research Center are the co-first authors.